This is big: EJS-ACT PD
This spring, your support can help accelerate the biggest ever clinical trial for Parkinson's and help us find better treatments, sooner. Donate today to bring hope to people with Parkinson’s and their loved ones.
£50 could pay for a specialised at-home biosample kit used to gather vital samples from trial participants.
- Paypal accepted
- Mastercard accepted
- Visa accepted
- American Express accepted
Find out more about EJS ACT-PD
EJS ACT-PD is the largest Parkinson’s clinical trial ever carried out in the UK, bringing together researchers to drive progress faster than ever before.
We urgently need to improve the speed and efficiency of trials because right now, someone in the UK is diagnosed every 20 minutes.
By 2050, the number of people living with Parkinson’s worldwide will double to 25 million.
It can take decades for treatments to reach people living with Parkinson’s. And existing therapies only manage symptoms. There is no current treatment that can stop its progression, but with your help, there's hope.
To improve the lives of people with Parkinson’s, we need to think big.
Why this trial is groundbreaking:
- EJS ACT-PD is a new style of trial, called multi-arm, multi-stage (MAMS). This means that, for the first time in Parkinson’s research, multiple treatments can be tested at the same time, giving us more answers quickly and efficiently
- We can check early on to see if treatments appear to be working. If they aren’t, they can be removed from the trial, and new ones can be added
- The drugs currently being tested in EJS ACT-PD are in phase 3 of clinical testing, the final hurdle before a treatment can be approved for use
- With 1,600 people with Parkinson’s across the UK taking part across 40 hubs, this is the UK’s biggest clinical research trial for Parkinson’s. This new type of trial has been highly effective for other conditions.
Trial lead, Professor Tom Foltynie, shared:
“When looking for new treatments for Parkinson’s, what frustrates everyone is the length of time it takes just to assess 1 treatment. Currently, treatments are tested one at a time, with each drug going through different clinical phases.’’
The EJS ACT-PD trial is set to change that.
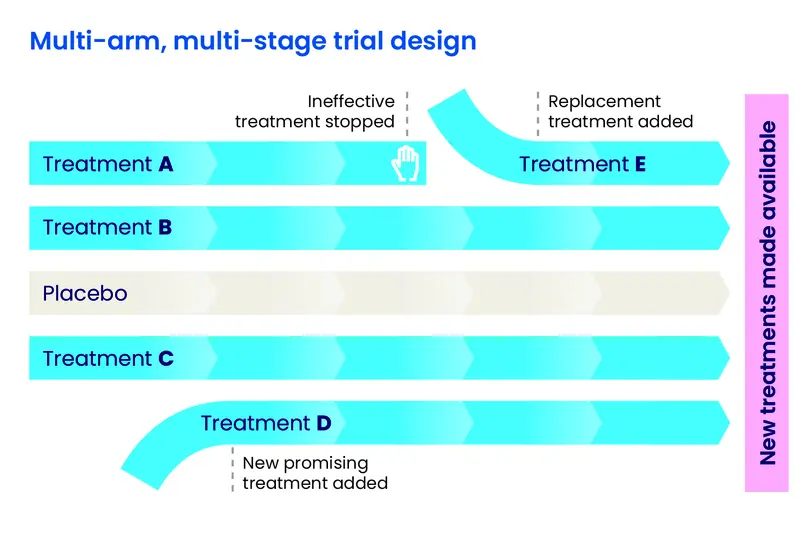
The platform trial approach
This approach has been used for other conditions including Multiple Sclerosis and prostate cancer. For example, the STAMPEDE clinical trial assessed new treatment approaches for people affected by high-risk prostate cancer.
It has led to finding new combinations of treatments that improve the overall survival rate. Now, we’re using this approach for Parkinson’s. With over 90 scientists from across the world coming together to establish EJS ACT-PD, we hope to find better treatments, faster.
The EJS ACT-PD trial is currently in its third and final phase of clinical testing. By focusing on repurposed drugs - medications already proven safe for other conditions - we’re cutting years off the traditional development timeline.
Led by experts, driven by patients
The trial is led by Tom Foltynie, Professor of Neurology at University College London, and Camille Carroll, Professor of Clinical Neuroscience at Newcastle University.
They’re part of a powerful global movement of scientists and innovators working to find new ways to slow and stop Parkinson’s. Together, they ensure that the perspective of people with Parkinson’s is included in every single decision. The trial continues to focus on meeting the needs of people with Parkinson’s and their loved ones.
Big or small, your contribution could change a life
Every donation is an investment into a future where Parkinson’s can be stopped or slowed, helping us to continue testing, learning, and developing the treatments that will change lives.
Together, we can give people living with Parkinson’s hope for a brighter future.
Or choose a specific project to support
Alongside the search for better treatments and a cure, we're addressing the everyday challenges of living with Parkinson's through non-drug solutions.
There is no approved treatment for Parkinson's hallucinations. Professor Suzanne Reeves and her team are investigating if an existing drug may be the answer.
The Parkinson's UK Brain Bank was established in 1984 at Imperial College London. Scientists around the world access the Brain Bank to deepen their understanding of the condition whilst searching for better treatments and ultimately a cure.
The Edmond J Safra Accelerating Clinical Trials in Parkinson’s Disease (EJS ACT-PD) trial is funded by a Medical Research Council (MRC) and National Institute for Health and Care Research (NIHR) partnership, Cure Parkinson’s, The Michael J Fox Foundation, Parkinson’s UK, The John Black Foundation, The Gatsby Charitable Foundation and Van Andel Institute.

